Research
EBMA’s research department, under the direction of Dr. Narges BAHI, conducts scientific evaluation programs for BI(G)MED. To achieve this, it focuses on 3 key areas.
1. Characterization of Ultra-Low Dose Solutions
One of our BI(G)MED products was subjected to an independent test conducted by the BION research laboratory, specializing in the analysis of subtle influences on water.
The test, which measures objective parameters such as pH, conductivity, and redox potential, showed a significant effect of the formula on water structuring.
This result is important: it confirms that our preparations, designed to act through fine and biomimetic modulation, produce a measurable and reproducible signal, consistent with our scientific approach.
In other words, this test provides external validation of the ability of BI(G)MED products to transmit regulatory information, thereby supporting the relevance of our innovative approach in bio-immunogenetic medicine.

2. In-Vitro Studies
A number of studies, whose results have not yet been published, have been conducted to evaluate the effect of our products on cultured cells. They show an effect on gene expression and cell function in culture.
3. Synthesis of Clinical Cases
Thanks to the entire network of BI(G)MED therapists, we are setting up retrospective studies on patients who have used BI(G)MED. These studies allow us to evaluate the real effectiveness of our formulas.
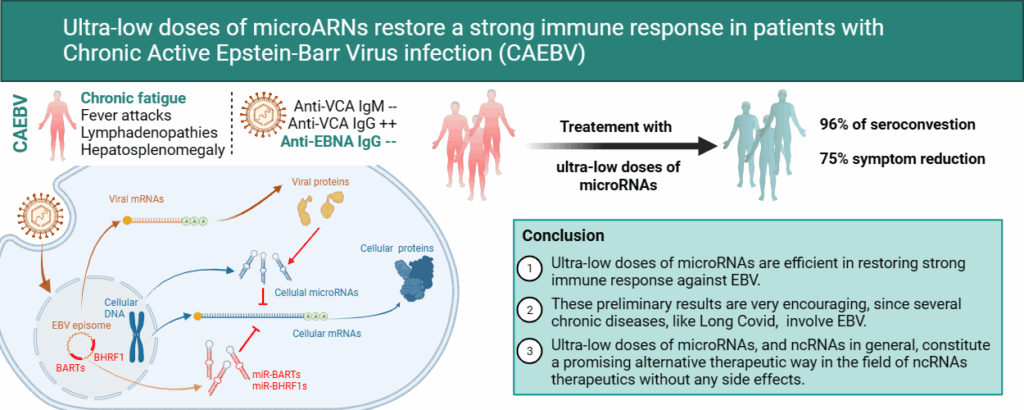
The latest retrospective study, published in the peer-reviewed International Journal of Scholarly Research and Reviews, focuses on patients with chronic mononucleosis (Epstein-Barr Virus (EBV) infection) characterized by the absence of anti-VCA IgM, the presence of anti-VCA IgG, and the absence of anti-EBNA IgG. This serological profile, characteristic of an incomplete immune response to EBV, shows a strong association with a number of chronic pathologies such as myalgic encephalomyelitis (chronic fatigue), certain autoimmune diseases, and even some cancers.
The objective of this study was to investigate the kinetics of seroconversion (i.e., development of anti-EBNA IgG) in patients treated with BI(G)MED.
The study results are conclusive: 95% of patients developed anti-EBNA IgG within an average of 7 months following the start of treatment, and this seroconversion was accompanied in 75% of cases by a marked improvement, or even complete disappearance, of their physical symptoms.
We are open to any partnership proposal to develop a randomized, double-blind clinical study on one or more of our products.
If you are interested, please contact our Scientific Director, Dr. Narges BAHI.